The Atom
Matter \(\Rightarrow\) Atoms \(\Rightarrow\) electrons, protons, and neutrons (except normal hydrogen does not have a neutron )
Periodic table \(\Rightarrow\) Elements \(\Rightarrow\) unique atomic structure
All atoms within a given element have the same number of protons.
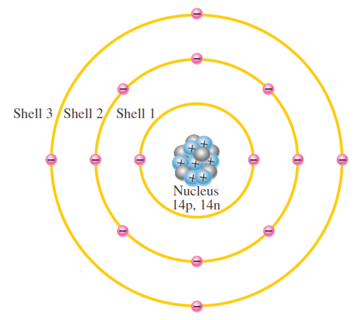
Atom is not a single particle but made up of small dense nucleus around which electrons orbit at a greater distance
Niels Bohr model \(\Rightarrow\) Planetary model \(\Rightarrow\) electrons in an atom circle the nucleus in different orbits similar to solar system
Quantum model is a more accurate representation, but difficult to visualize
For most practical purposes in electronics, the Bohr model suffices and is commonly used because it is easy to visualize.
The Bohr Model
Each of the known 118 elements has unique atomic structure
Protons : positively charged
Neutrons : uncharged particles
Electrons : negatively charged